Suvorova V.A., Pertsov N.B., Kotelnikov A.R. Sr-containing float slime ceramics - the final product of biomineral interaction.
The aim of the work has been to synthesize a matrix material for binding strontium contained in a wet float slime being a product of biocolloidal technology of radionuclides extraction from polluted: contaminated waters. Based on a study of strontium leaching, the possibility has been estimated for using solid model compositions produced by ceramization of Sr salts-kaolinites mixtures for a long-term immobilization of 87Sr under controlled storage.
Liquid radioactive waste (RAW) left after a fuel cycle or formed in emergency are very dangerous when stored. By joint efforts Russian and Ukrainian scientists developed a method for removing radionuclides from polluted waters accumulated under the failed reactor of the fourth block of the Chernobyl AES.
The method is based on treating the water by specially selected metalphilous micro-organisms with later separation of the disperse phase (radionuclide concentrate) by floatation. A decrease of the potential danger of RAWs and of their volume is reached by compacting, i.e. solidification. We have attempted to produce a solid alumosilicate matrix from Sr-containing float slimes, i.e. products of micro-organisms floatation, through their ceramization. To this end we used a sample of wet float slime containing about 10 wt% Sr kindly given to us by the colleagues from the Institute of biocolloidal chemistry of the Ukrainian NAS where it was produced by 1-2 intermixing of active slime (micro-organisms) with a water solution of strontium chloride (200 mg/l). Strontium was bound living cells united to flocules measuring 50-100 μm.
The separation of the Sr-loaded biomass was carried out using coagulants. 50 ml of the starting float slime was dried at 110oC until a solid residue was obtained, after that the residue was burnt at 700oC. The dry residue weight was 226 mg. A microprobe analysis of the powder showed that along with oxides of silicon (4.68), aluminium (7.43), calcium (2.39), high concentration of SrO (48.12%), it contains a large amount of Cl (21.03%).
The latter implied that strontium is retained as insoluble chloride, therefore it cannot be introduced into the alumosilicate matrix which is recognized as the most stable in keeping alkali-earth elements. For ceramization strontium ought to be in the form of compounds which easily decompose at high temperatures, i.e. carbonates or nitrates. The slime treatment with nitric acid yielded a chlorine-free product, but its amount was small and insufficient for conducting further runs. We, therefore, performed model experiments on introduction of strontium into kaolinite.
The introduction of strontium was performed by way of ceramization of calcinate from SrNO3 or SrCO3 and natural kaolinites with the addition of 1 wt% water in sealed platinum capsules at temperatures 700 and 1100oC and pressure 1000 atm. The initial charge was produced by a prolong
crushing and then drying at 110oC of kaolinite-salt mixture. The calcination of the initial charge was performed in an open crucible at 700oC for 1-3 days. In the table are listed to conditions and the results of the runs, the formulae are derived from the microprobe analysis data.
|
Run |
Initial charge |
Ň, 0Ń |
Đ, atm |
days |
Products |
d, g/cm3 |
P, % |
|
S-5 |
SrCO3 + kaolinite |
700 |
1000 |
3 |
(Na0.007K0,055Sr0.297)0.357(Al2.129Si2.239)4.368O24 |
2.7 |
12 |
|
S-6 |
SrCO3 + kaolinite |
1100 |
1000 |
3 |
(Na0.008K0,058Sr0.902)1.068(Al2.182Si2.114)4.297O24 |
2.3 |
23 |
|
S-7 |
SrCO3 + Kaol.Wool |
1100 |
1000 |
3 |
(Na0.038K0,009Sr0.787)0.834(Al2.060Si2.037)4.197O24 |
2.4 |
20 |
|
S-8 |
SrNO3 + Kaol.Wool |
1100 |
1000 |
3 |
(Na0.023K0,007Sr1.015)1.057(Al2.070Si1.982)4.042O24 |
2.7 |
20 |
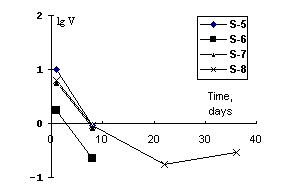
After quenching the run products were compact cylinders 30-40 mm length and 3-5 mm in dia. The X-ray patterns of the products exhibited reflexes of strontium feldspar. The ceramics quality was estimated by leaching of the elements out of the samples in distilled water at 90oC (IAAE test MCC-1). The results on strontium leaching are illustrated in the plot in the co-ordinates velocity lgV (g/m2.day) duration (day).
The results suggest that the charge from SrNO3 and kaoline wool gives the best ceramics. As little as 0.86% of strontium goes to the leaching medium in the course of the runs. The mean leaching velocity value, reached after 22 days, is 0.2 g/m2.day.
Considerable leaching velocities of Na 2.4 g/m2.day on the 40th day attest to a weak stability of this matrix material in general.
The leaching data obtained for strontium-containing ceramics on the base of the charge stoichiometrically corresponding to strontium anorthite showed that after the 40-day duration a mean leaching velocity of strontium, was 0.2 (lgV=-0.5) g/m2 that was better by a factor of 2 than from borosilicate glasses [1] with the initial strontium concentration being much higher in our samples. Despite the high porosity (~20%) dictates a rapid leaching of strontium and sodium, the sample retains a low leaching percent of Sr, that makes the use of our method promising for solidification of float slimes, that purify water basins, to the forms suitable for burial in rocks.

Reference: